相册共1张
点击图片可以查看相册
非洲绿猴肾细胞(Vero)
- 类型:原代细胞
- 品牌:Precision BioMedicals (PBM)
- 种属:African green monkey
规格: 1×10⁶ cells/T25瓶或2支冻存管
货号: A50180
价格: ¥960.00
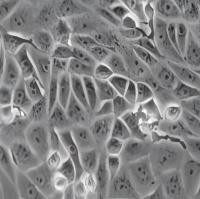
Vero细胞是日本千叶大学Yasumura Y 和Kawakita Y从正常成年非洲绿猴的肾脏组织中分离建立的。1964年6月15日,B. Simizu将其从千叶大学带到美国国立卫生研究院(NIH)过敏及传染病研究所热带病毒实验室时,已传至第93代。该细胞是贴壁依赖性的成纤维细胞,常作为转染宿主,也可用于支原体检测。此外,Vero细胞能支持多种病毒的增殖,包括乙型脑炎、脊髓灰质炎、狂犬病、新冠等病毒,已被准许用于生产人用病毒疫苗。
细胞质控检测
1) 支原体检测结果:阴性
2) 细菌检测结果:阴性
3) 真菌检测结果:阴性
4) STR鉴定结果:
- 该株细胞DNA进行猴源细胞STR分型结果显示,扩增后图谱清晰,分型结果良好;
- 该细胞为猴源Vero细胞,未发现存在人或其他猴源细胞污染。
- D8S1106 11 11
D1S518 15 16
DYS389 11 11
F13A01 17 26
D5S1467 10 10
D19S245 20 20
D6S1017 10 11
D17S1304 12 13
D4S2408 15 16
FGA 6 6
动物种别
非洲绿猴
组织来源
肾脏;疾病:正常细胞
形态
上皮细胞样,贴壁生长
培养基和添加剂配方
非洲绿猴肾细胞Vero完全培养液 配方:
参考传代条件
- 参考传代比例:1:3~1:5
- 参考传代周期:2~3天
- 参考换液频率:每周2~3次
- 冻存液配方:完全培养液95%,DMSO 5%
- 培养条件:气相:空气95%,二氧化碳5%;温度:37摄氏度。
细胞运输方式
活细胞保温运输或冻存管干冰运输。
供应限制
仅供研究之用
REFERENCE
British Pharmacopoeia Commission Tests for microbial contamination. London, UK:British Pharmacopoeia Commission;British Pharmacopoeia Appendix XVI B, 2003 Didier ES, et al. Characterization of Encephalitozoon (Septata) intestinailis isolates cultured from nasal mucosa and bronchoalveolar lavage fluids of two AIDS patients. J. Eukaryot. Microbiol. 43: 34-43, 1996. PubMed: 8563708 American Public Health Association. Compendium of methods for the microbiological examination of foods. 3rd ed.Washington, DC: American Public Health Association; 1992. Yasumura Y, Kawakita Y. Studies on SV40 in tissue culture - preliminary step for cancer research in vitro. Nihon Rinsho 21: 1201-1215, 1963. Simizu B, et al. Characterization of the Tacaribe group of arboviruses. I. Propagation and plaque assay of Tacaribe virus in a line of African green monkey kidney cells (Vero). Proc. Soc. Exp. Biol. Med. 125: 119-123, 1967. PubMed: 6027511 Rhim JS, Schell K. Cytopathic and plaque assay of rubella virus in a line of African green monkey kiency cells (Vero). Proc. Soc. Exp. Biol. Med. 125: 602-606, 1967. PubMed: 4961492 Liebhaber H, et al. Replication of rubella virus in a continuous line of African green monkey kidney cells (Vero). Proc. Soc. Exp. Biol. Med. 125: 636-643, 1967. PubMed: 4961494 Sasaki K, et al. Studies on measles virus. II. Propagation in two established simian renal cell lines and development of a plaque assay. Kitasato Arch. Exp. Med. 37: 27-42, 1964. PubMed:5833688 Earley E, et al. A plaque neutralization method for arboviruses. Proc. Soc. Exp. Biol. Med. 125(3): 741-747, 1967. PubMed:15938255 Rhim JS, et al. Temperature dependence of the synthesis of adenovirus tumor and viral antigens. Proc. Soc. Exp. Biol. Med. 127: 642-646, 1968. PubMed: 5689485 Rhim JS, Schell K. Cytopathic effects of the parainfluenza virus SV5 in Vero cells. Nature 216: 271-272, 1967. PubMed: 4293683 Ozawa Y. Studies on the replication of African horse-sickness virus in two different cell line cultures. Arch. Gesamte Virusforsch. 21: 155-169, 1967. PubMed: 4232530 Rhim JS, et al. Growth of Junin virus, the etiological agent of Argentinian hemorrhagic fever, in cell cultures. Arch. Gesamte Virusforsch. 21: 243-252, 1967. PubMed: 5591575 Huber M, et al. Tyrosine phosphorylation events during coxsackievirus B3 replication. J. Virol. 71: 595-600, 1997. PubMed: 8985388 Pugachev KV, et al. Improvement of the specific infectivity of the rubella virus (RUB) infectious clone: determinants of cytopathogenicity induced by RUB map to the nonstructural proteins. J. Virol. 71: 562-568, 1997. PubMed: 8985384 Mundt W, et al. Perfusion system and a method for the large scale production of virus or virus antigen. US Patent 5,719,051 dated Feb 17 1998 Nichol PF, et al. Herpes simplex virus gene expression in neurons: viral DNA synthesis is a critical regulatory event in the branch point between the lytic and latent pathways. J. Virol. 70: 5476-5486, 1996. PubMed: 8764059 Govorkova EA, et al. African green monkey kidney (Vero) cells provide an alternative host cell system for influenza A and B viruses. J. Virol. 70: 5519-5524, 1996. PubMed: 8764064 White LJ, et al. Attachment and entry of recombinant norwalk virus capsids to cultured human and animal cell lines. J. Virol. 70: 6589-6597, 1996. PubMed: 8794293 Martinez R, et al. Herpes simplex virus type 1 alkaline nuclease is required for efficient processing of viral DNA replication intermediates. J. Virol. 70: 2075-2085, 1996. PubMed: 8642627 Zeng L, et al. Identification of amino acids involved in a recognition by dengue virus NS3-specific, HLA-DR15-restricted cytotoxic CD4+ T-cell clones. J. Virol. 70: 3108-3117, 1996. PubMed: 8627790 Hill JM, et al. In vivo epinephrine reactivation of ocular herpes simplex virus type 1 in the rabbit is correlated to a 370-base-pair region located between the promoter and the 5' end of the 2.0-kilobase latency-associated transcript. J. Virol. 70: 7270-7274, 1996. PubMed: 8794381 Carter KL, et al. Characterization of the products of the UL43 gene of herpes simplex virus 1: potential implications for regulation of gene expression by antisense transcription. J. Virol. 70: 7663-7668, 1996. PubMed: 8892886 Malik AK, Weller SK. Use of transdominant mutants of the origin-binding protein (UL9) of herpes simplex virus type 1 to define functional domains. J. Virol. 70: 7859-7866, 1996. PubMed:8892908 Chen Y, et al. Demonstration of binding of dengue virus envelope protein to target cells. J. Virol. 70: 8765-8772, 1996. PubMed:8971005 Sandri-Goldin RM, Hibbard MK. The herpes simplex virus type 1 regulatory protein ICP27 coimmunoprecipitates with anti-sm antiserum, and the C terminus appears to be required for this interaction. J. Virol. 70: 108-118, 1996. PubMed: 8523514 Carter KL, Roizman B. The promoter and transcriptional unit of a novel herpes simplex virus 1 alpha gene are contained in, and encode a protein in frame with, the open reading frame of the alpha22 gene. J. Virol. 70: 172-178, 1996. PubMed: 8523523 Russell DW, Miller AD. Foamy virus vectors. J. Virol. 70: 217-222, 1996. PubMed: 8523528 Lukonis CJ, Weller SK. Characterization of nuclear structures in cells infected with herpes simplex virus type 1 in the absence of viral DNA replication. J. Virol. 70: 1751-1758, 1996. PubMed:8627697 Lagunoff M, et al. Phenotypic properties of herpes simplex virus 1 containing a derepressed open reading frame P gene. J. Virol. 70: 1810-1817, 1996. PubMed: 8627705 Uprichard SL, Knipe DM. Herpes simplex ICP27 mutant viruses exhibit reduced expression of specific DNA replication genes. J. Virol. 70: 1969-1980, 1996. PubMed: 8627723 Leopardi R, Roizman B. The herpes simplex virus major regulatory protein ICP4 blocks apoptosis induced by the virus or by hyperthermia. Proc. Natl. Acad. Sci. USA 93: 9583-9587, 1996. PubMed: 8790373 Pereira ME, et al. Invasive phenotype of Trypanosoma cruzi restricted to a population expressing trans-sialidase. Infect. Immun. 64: 3884-3892, 1996. PubMed: 8751943 AOAC International Virus in beef (ground), microbiological method. Gaithersburg, MD: AOAC International; AOAC "Official Methods of Analysis of the AOAC International" 975.56. Nucleofection technology from Lonza Single-use syringes (sterile) for the injection of 100 units per millilitre insulin (U-100), Appendix O. Sydney, NSW, Australia:Standards Australia;Standards Australia AS 1077-1992. Single-use(sterile) infusion sets for general medical use. Appendix F, Method of test for cytotoxicity. Sydney, NSW, Australia:Standards Australia;Standards Australia AS 2385-1990. Medical equipment--Single-use winged intravenous devices (sterile) for general medical use. Appendix J. Method of test for cytotoxicity. Sydney, NSW, Australia:Standards Australia;Standards Australia AS/NZS 2485:1995. Medical devices--Polymer urethral catherters for general medical use. Appendix B. Method of testing catheters for cytotoxicity. Sydney, NSW, Australia:Standards Australia;Standards Australia AS/NZS 2696:1996. General requirements for single-use, sterile, plasticized polyvinyl chloride (PVC) packs for human blood. Part2: Multiple blood pack systems. Appendix Q. Method of test for Cytotoxicity. Sydney, NSW, Australia:Standards Australia;Standards Australia AS 3787.2-1997. Testing of products for use in contact with drinking water. Sydney, NSW, Australia:Standards Australia;Standards Australia AS/NZS 4020:2002. Biological evaluation of medical devices. Part 5: Tests for in vitro cytotoxicity. Sydney, NSW, Australia:Standards Australia;Standards Australia AS ISO 10993.5-2002. Biological evaluation of medical devices--Part 5: Tests for in vitro cytotoxicity. Geneva (Switzerland):International Organization for Standardization/ANSI;ISO ISO 10993-5:1999. Test for absence of mycoplasmas. London, UK:British Pharmacopoeia Commission;British Pharmacopoeia Appendix XVI (Vet) 3, 2003 Standard Practice for Indirect Detection of Mycoplasma in Cell Culture by 4-6-Diamidino-2-2 Phenylindole (DAPI) Staining. West Conshohocken, PA:ASTM International;ASTM Standard Test Method E 1533-00 (Reapproved 2006). Standard Test Method for Determining the Virus-Eliminating Effectiveness of Liquid Hygienic Handwash and Handrub Agents Using the Fingerpads of Adult Volunteers. West Conshohocken, PA:ASTM International;ASTM Standard Test Method E 1838-02. Standard Quantitative Disk Carrier Test Method for Determining the Bactericidal, Virucidal, Fungicidal, Mycobactericidal and Sporicidal Activities of Liquid Chemical Germicides. West Conshohocken, PA:ASTM International;ASTM Standard Test Method E 2197-02.